Core Passerida
We have reached the heart of the Passerida, comprising over 35% of all extant bird species. We have also hit a major division, the division into Sylvioidea, the Muscicapoidea clade (including the former Bombycilloidea and Certhiodea), and Passeroidea.
Recent History of Top-Level Passerida Phylogeny: Over the last 2 decades, our understanding of Passerida's structure has undergone a revolution. I believe that revolution is now nearly complete. The publication by Oliveros et al. (2019), which built on Moyle et al. (2016), Kuhl et al. (2021), and Stiller et al. (2024) have answered most outstanding questions, although there's still uncertainty regarding the placement of some families in the Muscicapoidea.
The path that brought us here involved Barker et al. (2004), Barker (2014), and Ericson et al. (2014), who found that Paroidea and Sylvioidea group together, as do Muscicapoidea and Passeroidea. Barker (2014) grouped Certhiodea with Muscicapoidea and, with low support, grouped both Bombycilloidea and Reguloidea with Passeroidea. Both Irestedt and Ohlson (2008) and Johansson et al.'s (2008b) 6-gene analysis found Passeroidea to be the basal group, followed by Muscicapoidea, with Paroidea then sister to Sylvioidea. Alström et al.'s (2014) 7-gene analysis found a similar arrangment, with Reguloidea on its own branch. Johansson et al.'s (2008b) 3-gene analysis using more taxa came up with a different arrangement that also had Passeroidea basal. Hackett et al. (2008) only included a few taxa, but obtained a different arrangement with Bombycilloidea and Reguloidea sister to Muscicapoidea and Sylvioidea, and Passeroidea sister to the whole lot! Finally, Nabholz et al. (2011) argued in favor of a basal Muscicapoidea, with Paroidea the next branch, and Sylvioidea and Passeroidea as sisters.
At the top level, there are several basal Passerida families. These are followed by the Sylvioidea branch, which includes the former Paroidea. The remainder conveniently splits into an expanded Muscicapoidea (including the former Bombycilloidea, Reguloidea, and Certhioidea) and Passeroidea. The family phylogeny of both Sylvioidea and Passeroidea is close to settled. Muscicapoidea needs some more work.
Sylvioidea
Sylvioidea has been a mess for a long time. Both the Sylviidae and Timaliidae were treated as taxonomic trashcans (as were the Muscicapidae), and the boundaries between the two families have sometimes been rather vague. Species that were hard to classify got dumped in one or the other. This has been a particular problem because many of the species in Sylvioidea look very similar. The lack of a distinct juvenile plumage helps separate them from the Muscicapoidea, but in many cases there's really not much to go on in terms of morphology.
Just how much of a mess the Sylvioidea were is exemplified by the Sibley-Monroe checklist. They started with the Sylviidae, removed the cisticolas, kinglets, parids, and white-eyes and others, cleared out most of the corvid radiation, and still had a mass of 560 species lumped together in the family Sylviidae. The Timaliidae were nested inside it as a tribe (Timaliini). This was actually progress as it was an admission about how little we knew. In fact, the cisticolas, kinglets, parids, and white-eyes end up in the broader Sylvioidea, but not the family Sylviidae.
This new Sylvioidea underwent major restructuring, starting with the formation of a new superfamily---Certhioidea (Cracraft et al., 2004). The Certhioidea are comprised of several families of small birds that the modified Sibley-Ahlquist-Monroe list has at the beginning of the Sylvioidea. In fact, they are more closely related to the Muscicapoidea. One could even include them in the Muscicapoidea. So far, I've considered it better to place the Certhioidea in a separate superfamily. The families in question are the Sittidae (nuthatches), Tichodromidae (wallcreeper), Certhiidae (treecreepers), Polioptilidae (gnatcatchers and gnatwrens), and Troglodytidae (wrens).
Although these results cleared the ground, what was really needed was to sample a large number of sylvioid and timaliid taxa. Alström, Beresford, Ericson and others attacked the problem on a wide scale, while Cibois and company focused on Timaliidae. This process culminated in the paper by Oliveros et al. (2019), which presented a family level phylogeny for all of the Passeriformes. In the process, the Sylvioidea had been sliced and diced.
There is still work to be done on sylvioid and timaliid taxonomy, but we now have a pretty decent framework of sylvioid families. The framework has required some adjustment and rearrangement. Recent studies support the arrangement below. The framework can now be solidly based on three large-scale studies. The “big three” are Oliveros et al. (2019), Kuhl et al. (2021) and Stiller et al. (2024). The coverage of the Sylvioidea by Kuhl et al. and Stiller et al. is not entirely complete, but they include most of the sylvioid taxa and are based on a lot more genetic data for those they include.
There's a high degree of similarity between the big three phylogenies. Besides the missing taxa, the main differences are that Kuhl et al. place the Cisticolidae sister to Donacobiidae-Bernieridae-Locustellidae and Stiller et al. have the Pnoepygidae sister to the Hirundinidae.
The revised Sylvioidea required the creation of at fifteen new families: Hyliotidae, Stenostiridae, Panuridae, Nicatoridae, Macrosphenidae, Pnoepygidae, Acrocephalidae, Donacobiidae, Bernieridae, Locustellidae, Hyliidae, Cettiidae, Phylloscopidae, Pellorneidae, and Leiothrichidae.
One way to think of the new Sylvioidea is that it is babblers, bulbuls, larks, swallows, tits, and Old World warblers. All told, this adds to about one-eighth of all bird species.
The new Sylvioidea is very much an Old World group. Only the tits and swallows make it to the New World in numbers. Other than that, Sylvioidea contains only a small number of New World native breeders—the cosmopolitan Horned Lark (Alaudidae), the Neotropical Black-capped Donacobius (Donacobiidae), the Bushtit (Aegithalidae) and Verdin (Remizidae) of western North America, Alaska's Arctic Warbler (Phylloscopidae), and the Wrentit (Paradoxornithidae), which resides in far western North America, from near the mouth of the Columbia River to northern Baja California.
| Sylvioidea family tree |
|---|
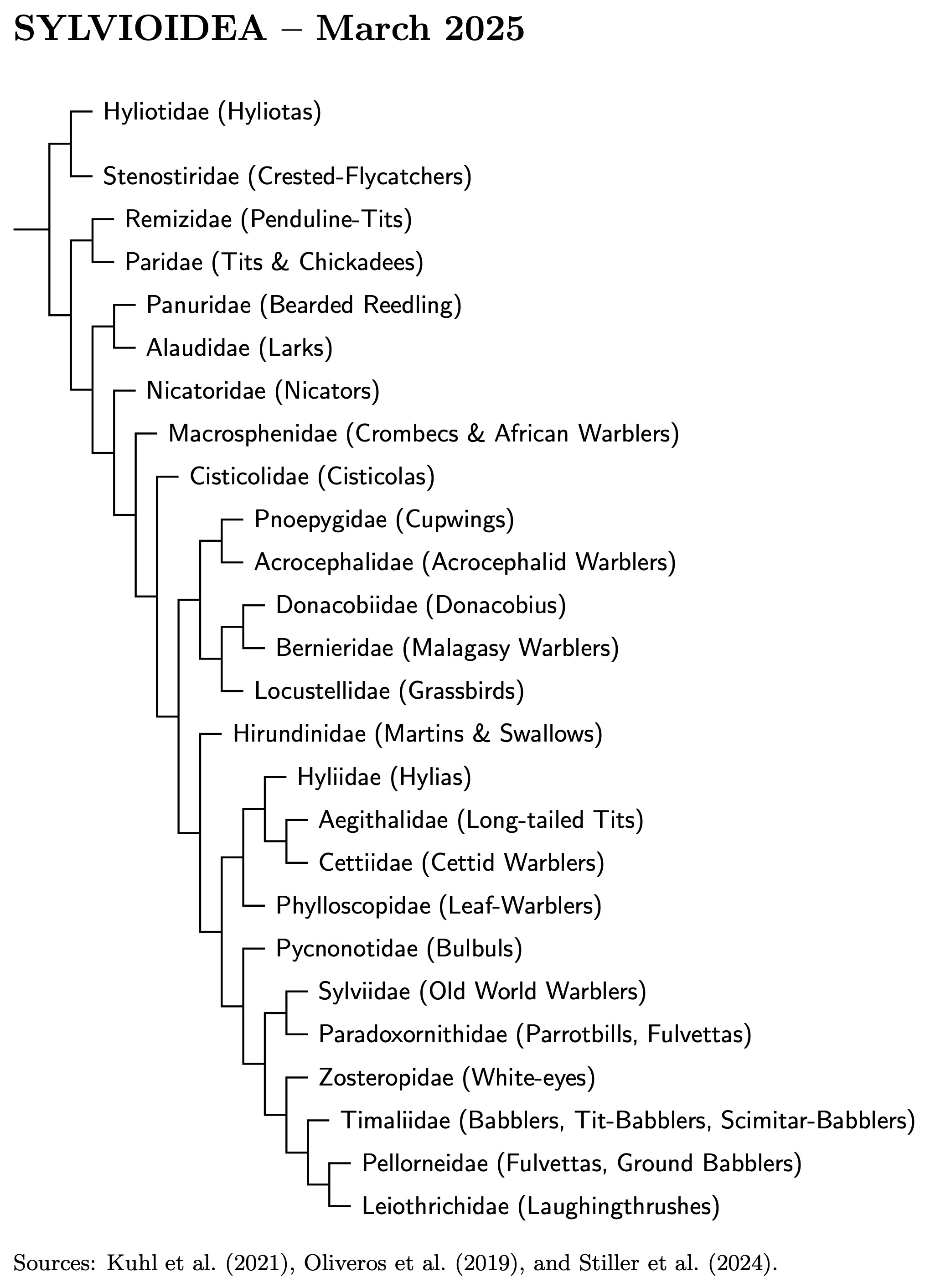 |
Those who look closely will see that families formerly in Paroidea are now the first four Sylvioidea. The problem was that the four Syvioidea families do not form a clade. Since I couldn't leave them all in Paroidea, it then seemed most natural to include them all in Sylvioidea.
The big three have given us great coverage of the Sylvioid famiies. All of the Sylvioid families are included in Oliveros et al., and most are also included in both Kuhl et al. (2021) and Stiller et al.
Remaining Conflicts: There are two important conflicts among the big three. The first involves the Cisticolidae. Oliveros et al. (2019) and Stiller et al. (2024) agree with the diagram, but Kuhl et al. (2021) considered the Cisticolidae sister to the Locustellidae-Donacobiidae-Berneiridae clade.
The second conflict concerns the cupwings (Pnoepygidae). Kuhl et al. have it sister to the reed warblers (Acrocephalidae), as on the diagram. In contrast, it is sister to the swallows (Hirundininae) in Oliveros et al. They are not included in Stiller et al.
For those wanting more information on the history of the new Sylvioidea, I recommend starting with Don Roberson's web page: The Break-up of the Old World Warblers, It covers the changes to the Sylviidae that started the process. For the gory details, the papers by Alström et al. (2006), Barker et al. (2004), Beresford et al. (2005), Jønsson and Fjeldså (2006a), Johansson et al. (2008b), Gelang et al. (2009), and Fregin et al. (2012) lay out the big picture, and Cibois (2003), Cibois et al. (1999, 2001, 2002), Fuchs et al. (2006a, b), Nguembock et al. (2007), Pasquet et al. (2006), and Zhang et al. (2007) are helpful.
Hyliotas and Fairy Flycatchers
Hyliotidae: Hyliotas Fjeldså, 2014 (H&M-4)
1 genus, 4 species Not HBW Family
This small group of African endemics was considered part of Sylviidae until recently. Current evidence suggests they are not particularly closely related to Sylviidae. Rather, together with the Stenostiridae they form the basal branch of the larger Sylvioidea.
I think that Hyliotidae is allowed as a name. However, there is a complication. Hyliotinae was used by Reitter in 1880 for a beetle subfamily (now called Brontinae). It was based on the preocupied beetle genus Hyliota Reitter 1880 even though the bird genus Hyliota Swainson 1827 has priority over Hyliota. I'm not sure exactly how ICZN rules apply to a case like this. I don't know whether Hyliotidae can legitimately be used for this group without an ICZN ruling.
- Yellow-bellied Hyliota, Hyliota flavigaster
- Southern Hyliota, Hyliota australis
- Usambara Hyliota, Hyliota usambara
- Violet-backed Hyliota, Hyliota violacea
Stenostiridae: Fairy Flycatchers Fuchs et al., 2009
4 genera, 9 species Not HBW Family
 |
| Click for Stenostiridae tree |
|---|
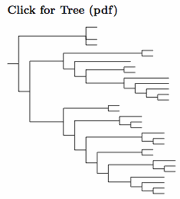
The Stenostiridae are a group of small African and Asian flycatchers that were previously scattered across three superfamilies but belong to none of them. The Stenostiridae are a new family. The name was used informally by Beresford et al. (2005), but the family was not formally named until Fuchs et al. (2009).
The Stenostiridae include the Fairy Flycatcher Stenostira (Sylvioidea: Sylviidae), the Yellow-bellied Flycatcher, Chelidorhynx hypoxanthus (Corvoidea: Rhipiduridae) Elminia blue-flycatchers (Corvoidea: Monarchidae), "Elminia" crested-flycatchers (also Corvoidea: Monarchidae), and the Culicicapa canary-flycatchers (Muscicapoidea: Muscicapidae). The three "Elminia" were previously considered part of the genus Trochocercus in the corvid family Monarchidae. Although they are sister to the true Elminia, their most recent common ancestor lived roughly 15 million years ago. They desperately need a new genus name. For now, I'm using the temporary name "Elminia".
Nguembock et al. (2008b) was the first paper to analyze the 8 then-known species in the Stenostiridae. Based on their chronogram, the crested ("Elminia") and blue (Elminia) flycatchers are the most closely related genera in the Stenostiridae. With 15 million separation, close is a relative term here. Nguembock et al. did not consider Chelidorhynx as it had not yet been placed in Stenostiridae. It seems likely its common ancestor with Stenostira lived about 20 mya, based on the analysis in Fuchs et al. (2009). These ages seem consistent with Oliveros et al. (2019). The Stenostiridae are each others closest relatives, even though some are not very close. It is a coherent enough group to call it a family, even after we bring in a bird thought to be a fantail!
The Fantail: Nyári et al. (2009a) and Fuchs et. al (2009) found a ninth member of the Stenostiridae. It is the Yellow-bellied Fantail, Rhipidura hypoxantha, which had been considered unusual. Some authors had previously separated it as the monotypic genus Chelidorhynx. Accordingly, it becomes Chelidorhynx hypoxanthus. Fuchs et al. (2009) show that it is sister to the Fairy Flycatcher. The arrangement of species follows Nguembock et al. (2008b) and Figures 3 and 4 of Fuchs et al. (2009).
Which Group Name for Elminia? I previously listed two English names for each of the true Elminia. I've now removed one set, the Crested-Flycatcher names, leaving the only the Blue Flycatcher names (not to be confused with the Cyornis Blue-Flycatchers).
- Yellow-bellied Flycatcher / Yellow-bellied Fantail, Chelidorhynx hypoxanthus
- Fairy Flycatcher, Stenostira scita
- Gray-headed Canary-flycatcher, Culicicapa ceylonensis
- Citrine Canary-flycatcher, Culicicapa helianthea
- African Blue Flycatcher, Elminia longicauda
- White-tailed Blue Flycatcher, Elminia albicauda
- Dusky Crested-flycatcher, "Elminia" nigromitrata
- White-bellied Crested-flycatcher, "Elminia" albiventris
- White-tailed Crested-flycatcher, "Elminia" albonotata
Tits and Penduline Tits
Remizidae: Penduline-Tits Olphe-Galliard, 1891
3 genera, 11 species HBW-13
 |
| Click for Remizidae tree |
|---|
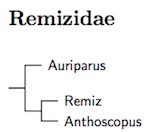
Although Cephalopyrus is usually placed in this family, the available molecular data put it in the Paridae (Tietze and Borthakur, 2012; Ball, 2014). Ball (2014) examined all the penduline tits. The arrangement here is based on Ball (2014) and Barani-Beiranvand et al. (2017). Ball included one sample from the subspecies taruensis in his analysis. As a result, there is a question whether taruensis should become a subspecies of musculus rather than caroli (where it is often subsumed in robertsi).
- Verdin, Auriparus flaviceps
- Chinese Penduline-Tit, Remiz consobrinus
- White-crowned Penduline-Tit, Remiz coronatus
- Eurasian Penduline-Tit, Remiz pendulinus
- Black-headed Penduline-Tit, Remiz macronyx
- Yellow Penduline-Tit, Anthoscopus parvulus
- Cape Penduline-Tit, Anthoscopus minutus
- Forest Penduline-Tit, Anthoscopus flavifrons
- Sennar Penduline-Tit, Anthoscopus punctifrons
- Gray Penduline-Tit, Anthoscopus caroli
- Mouse-colored Penduline-Tit, Anthoscopus musculus
Paridae: Tits, Chickadees, Titmice Vigors, 1825
14 genera, 66 species HBW-12
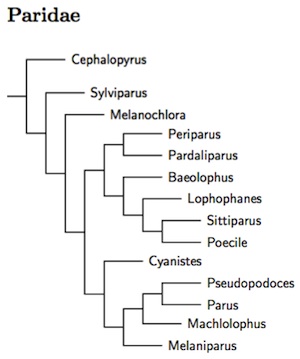
The two main changes in the membership of the Paridae have already been mentioned: the Paridae gain Hume's Groundpecker (formerly Hume's Ground-Jay), Pseudopodoces humilis from the Corvidae (see James et al., 2003; Gill et al., 2005) and also the Fire-capped Tit, Cephalopyrus flammiceps from the closely related Remizidae (Tietze and Borthakur, 2012). I had previously based the arrangement of genera and species on a combination of Dai et al. (2010), Gill et al. (2005), Illera et al. (2011), James et al. (2003), and Tietze and Borthakur (2012). The current arrangement follows the comprehensive multi-gene analysis (2 nuclear, 1 mitochondrial) of Johansson et al. (2013), which is similar to previous arrangement, but differs in detail. As most nodes in Johansson et al. were strongly supported, I have adopted it wholesale.
 |
| Click for Paridae tree |
|---|
The Paridae start with 3 basal species, the Fire-capped Tit (formerly in Remizidae), Yellow-browed Tit, and Sultan Tit. The remainder of the Paridae then divide into two clades. The first clade includes the coal tit and allies (Periparus and Pardaliparus), the titmice (Baeolophus), crested-tits (Lophophanes), the Varied Tit and White-fronted Tit (now united in Sittiparus), the Eurasian crested tits (Lophophanes), Nearctic crested tits (Baeolophus) and the chickadees (Poecile). The second clade includes the blue-tits (Cyanistes), Hume's Groundpecker (Pseudopodoces), the Great Tit and allies (Parus), the remaining Indo-Malayan tits (Machlolophus), and the Afrotropical tits (Melaniparus).
This involves the use of 4 additional genera, as recommened by Johansson et al. (2013). There are two in the first clade. These allows separating the Coal Tit and allies into Periparus (mostly temperate) from Pardaliparus (tropical and subtropical, Selys-Longchamps 1884, type elegans). The Varied and White-fronted Tits of the far east are separated as Sittiparus (Selys-Longchamps 1884, type varius). Further, based on McKay et al. (2014), the Chestnut-bellied Tit, Sittiparus castaneoventris, and Iriomote Tit, Sittiparus olivaceus, have been separated from Varied Tit, Sittiparus varius. The case for olivaceus is rather marginal. However, Owston's Tit, Sittiparus owstoni, was found to be embedded in the remaining portion of the varius complex and is not recognized, contra McKay et al. (2014).
The two additional genera in the second clade permit us to retain the distinctive Hume's Groundpecker in a separate genus. The former Parus tits from the Afrotropics are put in Melaniparus (Bonaparte 1850, type niger) while the Indo-Malayan tits go in Machlolophus (Cabanis 1851, type spilonotus).
Johansson et al. (2013) also found that the Caspian Tit, Poecile hyrcanus, is not closely related to the Sombre Tit, Poecile lugubris. As a result, the Caspian Tit is treated as a separate species here. The arrangement of the chickadees is based on Harris et al. (2014a). They sequenced a number of additional nuclear genes and found this to be the most likely topology. Mitochondrial genes consistently give a different result where the plumage does not make sense.
The African Blue Tit, Cyanistes teneriffae, has been split into Palma Blue Tit, Cyanistes palmensis, Libyan Blue Tit, Cyanistes cyrenaicae, Canary Blue Tit, Cyanistes teneriffae, and Ultramarine Tit, Cyanistes ultramarinus, based on Stervander et al. (2015). See also Illera et al. (2011), Päckert et al. (2013b), and Gohli et al. (2014). Given that two Cyanistes now have names other than “Blue Tit”, I have removed the hyphens.
Stervander et al. (2015) find evidence that three lineages of Blut Tits have invaded the Canary Island. The Palma and Libyan Blue Tits are relics of an earlier African lineage, while the Canary Blue Tit (C. teneriffae, including ombriosus and hedwigii) has separated from the more recent African lineage. Finally, its sister lineage, the Ultramarine Tit, C. ultramarinus, of northwest Africa has also invaded the Canaries in Fuerteventura and Lanzarote (subspecies degener).
The Turkestan Tit, Parus major bokharensis group, is treated as a subspecies of the Great Tit, Parus major, based on the analyses of Kvist et al. (2003) and Päckert et al. (2005). However, they studied very few individuals from the bokharensis group. Their results did not quite match (except that bokharensis is quite variable genetically) and the individual studied by Johansson et al. (2013) ended up on a longer than expected branch. Further investigation may support species status for the Turkestan Tit.
- Fire-capped Tit, Cephalopyrus flammiceps
- Yellow-browed Tit, Sylviparus modestus
- Sultan Tit, Melanochlora sultanea
- Coal Tit, Periparus ater
- Rufous-naped Tit, Periparus rufonuchalis
- Rufous-vented Tit, Periparus rubidiventris
- Yellow-bellied Tit, Pardaliparus venustulus
- Elegant Tit, Pardaliparus elegans
- Palawan Tit, Pardaliparus amabilis
- Bridled Titmouse, Baeolophus wollweberi
- Oak Titmouse, Baeolophus inornatus
- Juniper Titmouse, Baeolophus ridgwayi
- Tufted Titmouse, Baeolophus bicolor
- Black-crested Titmouse, Baeolophus atricristatus
- Gray Crested-Tit, Lophophanes dichrous
- Crested Tit / European Crested-Tit, Lophophanes cristatus
- White-fronted Tit, Sittiparus semilarvatus
- Chestnut-bellied Tit, Sittiparus castaneoventris
- Varied Tit, Sittiparus varius
- Iriomote Tit, Sittiparus olivaceus
- White-browed Tit, Poecile superciliosus
- Sombre Tit, Poecile lugubris
- Pere David's Tit, Poecile davidi
- Marsh Tit, Poecile palustris
- Caspian Tit, Poecile hyrcanus
- Black-bibbed Tit, Poecile hypermelaenus
- Willow Tit, Poecile montanus
- Sichuan Tit, Poecile weigoldicus
- Gray-headed Chickadee, Poecile cinctus
- Boreal Chickadee, Poecile hudsonicus
- Chestnut-backed Chickadee, Poecile rufescens
- Black-capped Chickadee, Poecile atricapillus
- Carolina Chickadee, Poecile carolinensis
- Mountain Chickadee, Poecile gambeli
- Mexican Chickadee, Poecile sclateri
- Eurasian Blue Tit, Cyanistes caeruleus
- Azure Tit, Cyanistes cyanus
- Palma Blue Tit, Cyanistes palmensis
- Libyan Blue Tit, Cyanistes cyrenaicae
- Canary Blue Tit, Cyanistes teneriffae
- Ultramarine Tit, Cyanistes ultramarinus
- Hume's Groundpecker / Ground Tit, Pseudopodoces humilis
- Green-backed Tit, Parus monticolus
- Great Tit, Parus major
- Japanese Tit, Parus minor
- Cinereous Tit, Parus cinereus
- White-naped Tit, Machlolophus nuchalis
- Yellow Tit, Machlolophus holsti
- Yellow-cheeked Tit, Machlolophus spilonotus
- Himalayan Black-lored Tit, Machlolophus xanthogenys
- Indian Black-lored Tit, Machlolophus aplonotus
- White-shouldered Black-Tit, Melaniparus guineensis
- White-winged Black-Tit, Melaniparus leucomelas
- Rufous-bellied Tit, Melaniparus rufiventris
- Cinnamon-breasted Tit, Melaniparus pallidiventris
- White-bellied Tit, Melaniparus albiventris
- Carp's Tit, Melaniparus carpi
- Southern Black-Tit, Melaniparus niger
- Dusky Tit, Melaniparus funereus
- Miombo Tit, Melaniparus griseiventris
- Stripe-breasted Tit, Melaniparus fasciiventer
- Acacia Tit, Melaniparus thruppi
- Red-throated Tit, Melaniparus fringillinus
- White-backed Black-Tit, Melaniparus leuconotus
- Ashy Tit, Melaniparus cinerascens
- Gray Tit, Melaniparus afer
Larks and Reedling
There have been many opinions about how to classify the Reedling, and it was sometimes considered a parid or parrotbill, usually with a name to match (Bearded Tit or Bearded Parrotbill). It had recently been considered a parrotbill (Paradoxornithidae).
Using overlapping but distinct data sets, Alström et al. (2006), Ericson and Johansson (2003), Fregin et al. (2012), and Fuchs et al. (2006a) found that Panurus is sister to the larks.
The big three (Oliveros, Kuhl, Stiller) agree. Kuhl et al. (2021) date the Panurus-Alaudidae split at 21.3 mya, Stiller et al. (2024) estimate it at 17.5 mya, and Oliveros et al. (2019) put it at about 21.1 mya. The age, and the fact that the Reedling is quite different from the larks, justify its treatment as a family.
Panuridae: Bearded Reedling des Murs, 1860
1 genus, 1 species Not HBW Family
- Bearded Reedling, Panurus biarmicus
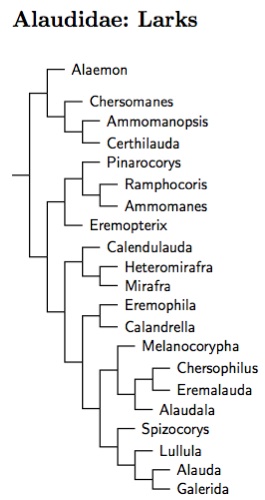
Alaudidae: Larks Vigors, 1825
21 genera, 100 species HBW-9
 |
| Click for Alaudidae tree |
|---|
The larks are the major piece of this clade. Although they were considered part of Passeroidea in the modified Sibley-Ahlquist-Monroe list, this is not correct. Rather, they belong in the Sylvioidea. Although the placement of the larks may have changed, their composition has not. This reflects the fact that they are one of the two easily identifiable passerine families (the swallows are the other).
Previously, Tieleman et al. (2003) found that some of the generic limits need adjusting (including Mirafra) and that some general reorganization is called for. Some of the blanks were filled in as Ryan et al. (1998) separated Barlow's Lark from Karoo Lark, while Ryan and Bloomer (1999) studied the long-billed larks. Gray's Lark is now in a different genus (Ammomanopsis), as are the long-billed larks (Certhilauda).
Note that the Sidamo Lark, Heteromirafra sidamoensis, has been lumped with Archer's Lark, Heteromirafra archeri. Spottiswoode et al. (2013) found virtually no genetic distance between them.
The comprehensive genetic analysis by Alström et al. (2013) has prompted additional adjustments of genetic boundaries and an overall reorganization of the family. They analyzed about 5/6 of the lark species. They found that Pseudalaemon is embedded in Spizocorys, that the Madagascan Lark, Mirafra hova, belongs in Eremopterix, that Calandrella is really two genera (Alaudala is the other), and that the White-winged Lark, Melanocorypha leucoptera, appears to belong in or near Alauda.
More precisely, the Sand Lark, and Athi, Asian, Lesser, and maybe Somali Short-toed Larks are not closely related to the other Calandrella larks. This group takes the name Alaudala (Horsfield and Moore 1856, type raytal).
The remaining Calandrella have been studied in more detail by Stervander et al. (2016). I use their arrangment, and have implemented three splits.
- Aden Lark, Calandrella eremica (including daaroodensis), of southwestern Arabia and northern Somalia has been split from Blanford's Lark, Calandrella blanfordi of northern Eritrea.
- Red-capped Lark, Calandrella cinerea, is split into Northern Red-capped Lark, Calandrella williamsi, and Southern Red-capped Lark, Calandrella cinerea. The Northern Red-capped Lark only the Kenyan birds (williamsi) and Nigerian birds usually attributed to saturatior. If they had a scientific name, I would probably split the Nigerian and Kenyan birds into two species. The type of saturatior is from Angola, representing a Southern Red-capped Lark. The isolated northern populations in the DR Congo and Uganda belong in Southern Red-capped Lark.
- Sykes's Short-toed Lark, Calandrella dukhunensis, has been split from Greater Short-toed Lark, Calandrella brachydactyla. The name Rufous Short-toed Lark has been used for this species, but has also been used more appropriately for the Somali Short-toed Lark, Alaudala somalica.
The issue of the White-winged Lark is interesting. Its plumage is very similar to the Mongolian Lark, Melanocorypha mongolica, and it is hard to see how such similar plumage would evolve independently. However, the genetic analysis was very clear that it is not in Melanocorypha, instead falling in a clade that includes Galerida and Alauda. Exactly where in the clade is not so clear. The combined analysis places it sister to Alauda, but it may be sister to Galerida plus Alauda. Alström et al. recommend placing it in Alauda, and I follow that here. But what about the plumage? It is not unknown for there to be errors in the lab, and I'm not convinced this has been completely ruled out. However, as Alström et al. point out, it does seem more like Alauda in behavior, ecology, morphology, and vocally, so maybe it is an Alauda. But then how did the White-winged and Mongolian Larks end up with such similar plumage? No other lark has such plumage.
Guillaumet et al. (2005, 2006, 2008) have been studying the Galerida larks. This has lead to the split of Crested Lark, Galerida cristata, with the long-billed subspecies in the Maghreb becoming Maghreb Lark, Galerida macrorhyncha, while the short-billed subpsecies remain in Galerida cristata. The subspecies of Thekla Lark, Galerida theklae, around the horn of Africa seem to be candidates for one or more future splits (the subspecies involved include ellioti, praetermissa, and hueii).
Although coverage of subspecies was limited, Alström et al. (2013) also found indications that the Thekla Lark contains multiple species. There may also be multiple species within the Desert Lark. Further, the Greater Short-toed Lark is also a candidate for a split.
Alström et al.'s (2013) results also suggest that there are multiple species within the Horned Lark complex. In fact, their results suggested that two of the races of Horned Lark appeared closer to Temminck's Lark, and the genetic distance between American Horned Larks and some (but not all) of the Eurasian Horned Larks was considerable. However, Drovetski et al. (2014) found small genetic distances between all of these taxa. Although they suggested treating the group as 6 species instead of 2, my own reading is that they are better treated as 4 subspecies groups. The Horned Larks then become 4 groups: longirostris (argalea, teleschowi, przewalskii, nigrifrons, deosaiensis, longirostris, elwesi, khamensis); Temminck's Lark, E. bilopha (monotypic); penicillata (atlas, balcanica, kumerloevei, penicillata, bicornis, albigula); and alpestris (flava, brandti, and all New World subspecies). I don't see why the allopatric Temminck's Lark should be treated as a separate species, but until I find out more about why it was split, I maintain the current treatment.
- Greater Hoopoe-Lark, Alaemon alaudipes
- Lesser Hoopoe-Lark, Alaemon hamertoni
- Spike-heeled Lark, Chersomanes albofasciata
- Beesley's Lark, Chersomanes beesleyi
- Gray's Lark, Ammomanopsis grayi
- Short-clawed Lark, Certhilauda chuana
- Benguela Long-billed Lark, Certhilauda benguelensis
- Karoo Long-billed Lark, Certhilauda subcoronata
- Eastern Long-billed Lark, Certhilauda semitorquata
- Cape Long-billed Lark, Certhilauda curvirostris
- Agulhas Long-billed Lark, Certhilauda brevirostris
- Rufous-rumped Lark, Pinarocorys erythropygia
- Dusky Lark, Pinarocorys nigricans
- Thick-billed Lark, Ramphocoris clotbey
- Bar-tailed Lark, Ammomanes cinctura
- Desert Lark, Ammomanes deserti
- Rufous-tailed Lark, Ammomanes phoenicura
- Black-eared Sparrow-Lark, Eremopterix australis
- Madagascan Lark, Eremopterix hova
- Black-crowned Sparrow-Lark, Eremopterix nigriceps
- Chestnut-backed Sparrow-Lark, Eremopterix leucotis
- Ashy-crowned Sparrow-Lark, Eremopterix griseus
- Chestnut-headed Sparrow-Lark, Eremopterix signatus
- Gray-backed Sparrow-Lark, Eremopterix verticalis
- Fischer's Sparrow-Lark, Eremopterix leucopareia
- Pink-breasted Lark, Calendulauda poecilosterna
- Foxy Lark, Calendulauda alopex
- Fawn-colored Lark, Calendulauda africanoides
- Sabota Lark, Calendulauda sabota
- Karoo Lark, Calendulauda albescens
- Red Lark, Calendulauda burra
- Dune Lark, Calendulauda erythrochlamys
- Barlow's Lark, Calendulauda barlowi
- Archer's Lark, Heteromirafra archeri
- Rudd's Lark, Heteromirafra ruddi
- Collared Lark, Mirafra collaris
- Rusty Bushlark, Mirafra rufa
- Gillett's Lark, Mirafra gilletti
- Eastern Clapper-Lark, Mirafra fasciolata
- Cape Clapper-Lark, Mirafra apiata
- Rufous-naped Lark, Mirafra africana
- Red-winged Lark, Mirafra hypermetra
- Somali Lark, Mirafra somalica
- Ash's Lark, Mirafra ashi
- Flappet Lark, Mirafra rufocinnamomea
- Angolan Lark, Mirafra angolensis
- Burmese Bushlark, Mirafra microptera
- Bengal Bushlark, Mirafra assamica
- Indian Bushlark, Mirafra erythroptera
- Jerdon's Bushlark, Mirafra affinis
- Indochinese Bushlark, Mirafra erythrocephala
- Williams's Lark, Mirafra williamsi
- Monotonous Lark, Mirafra passerina
- Melodious Lark, Mirafra cheniana
- Horsfield's Bushlark, Mirafra javanica
- Singing Bushlark, Mirafra cantillans
- Kordofan Lark, Mirafra cordofanica
- White-tailed Lark, Mirafra albicauda
- Friedmann's Lark, Mirafra pulpa
- Horned Lark, Eremophila alpestris
- Temminck's Lark, Eremophila bilopha
- Aden Lark, Calandrella eremica
- Blanford's Lark, Calandrella blanfordi
- Erlanger's Lark, Calandrella erlangeri
- Northern Red-capped Lark, Calandrella williamsi
- Southern Red-capped Lark, Calandrella cinerea
- Greater Short-toed Lark, Calandrella brachydactyla
- Hume's Short-toed Lark, Calandrella acutirostris
- Sykes's Short-toed Lark, Calandrella dukhunensis
- Bimaculated Lark, Melanocorypha bimaculata
- Calandra Lark, Melanocorypha calandra
- Black Lark, Melanocorypha yeltoniensis
- Tibetan Lark, Melanocorypha maxima
- Mongolian Lark, Melanocorypha mongolica
- Dupont's Lark, Chersophilus duponti
- Dunn's Lark, Eremalauda dunni
- Athi Short-toed Lark, Alaudala athensis
- Asian Short-toed Lark, Alaudala cheleensis
- Sand Lark, Alaudala raytal
- Lesser Short-toed Lark, Alaudala rufescens
- Somali Short-toed Lark, Alaudala somalica
- Stark's Lark, Spizocorys starki
- Sclater's Lark, Spizocorys sclateri
- Short-tailed Lark, Spizocorys fremantlii
- Masked Lark, Spizocorys personata
- Obbia Lark, Spizocorys obbiensis
- Pink-billed Lark, Spizocorys conirostris
- Botha's Lark, Spizocorys fringillaris
- Woodlark, Lullula arborea
- White-winged Lark, Alauda leucoptera
- Raso Lark, Alauda razae
- Eurasian Skylark, Alauda arvensis
- Oriental Skylark, Alauda gulgula
- Sun Lark, Galerida modesta
- Large-billed Lark, Galerida magnirostris
- Thekla Lark, Galerida theklae
- Crested Lark, Galerida cristata
- Maghreb Lark, Galerida macrorhyncha
- Malabar Lark, Galerida malabarica
- Sykes's Lark, Galerida deva
Nicatoridae and Macrosphenidae
The Nicatoridae and Macrosphenidae are two small independent branches in Sylvioidea.
Nicatoridae: Nicators Cracraft, Beresford, and Barker, 2014 (H&M-4)
1 genus, 3 species Not HBW Family
- Western Nicator, Nicator chloris
- Eastern Nicator, Nicator gularis
- Yellow-throated Nicator, Nicator vireo
The nicators form a small African clade. They were once considered to be a type of bulbul, but are not closely related to bulbuls or anything else (Beresford et al., 2005; Johansson et al., 2008b; Oliveros et al., 2019; Kuhl et al., 2021; Stiller et al., 2024). They are an independent branch in Sylvioidea, between the Paridae/Remizidae and Panuridae/Alaudidae branches. That means they require their own family. They were named by Fregin et al. (2012) who noted their connection to the Panuridae/Alaudidae clade.
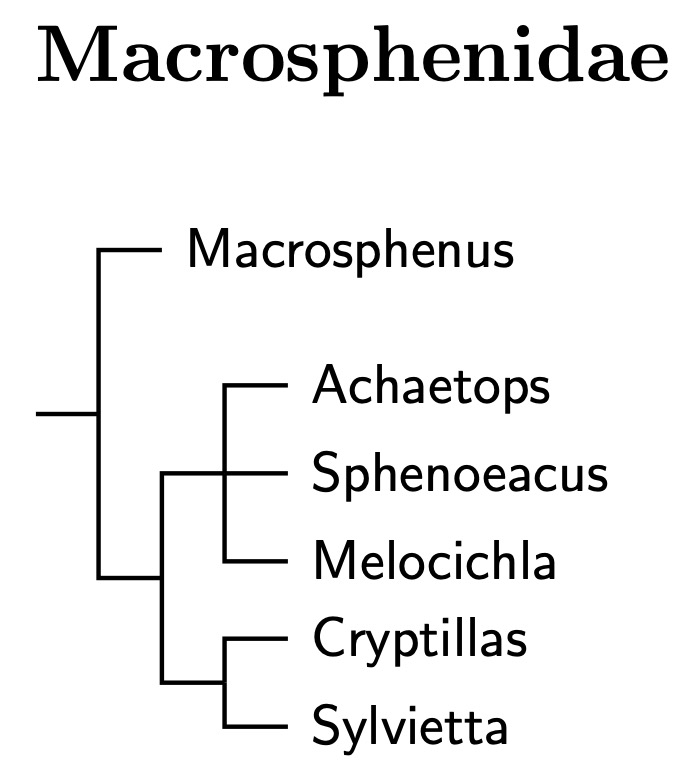
Macrosphenidae: Crombecs, African Warblers Fregin et al., 2012
6 genera, 18 species Not HBW Family
 |
| Click for Macrosphenidae tree |
|---|
The Macrosphenidae are another new family. This is formed entirely from the former Sylviidae and consists of crombecs and some African warblers (see Beresford et al., 2005; Nguembock et al., 2007). Johansson et al. (2008b) and Fregin et al. (2012) put it next in order. The big three concur.
Victorin's Warbler was formerly considered part of the genus Bradypterus. The rest of Bradypterus is now in Locustellidae.
Although Alström placed Macrosphenus kretschmeri in the bulbuls, this turned out to be a mistake based on a mislabelled specimen (Johansson et al., 2007b). However, Macrosphenus kretschmeri is different enough from the other Macrosphenus to sometimes get its own genus (Suaheliornis). It has yet to be sequenced and it is possible it does not belong here. The arrangement of Crombecs follows Huntley and Voelker (2017). The rest of the family has been arranged using Johansson et al. (2007b).
Huntley and Voelker (2017) estimated that the most recent common ancestor of Sylvietta lived around 6 mya. Oliveros et al. (2019) put the common ancestor of Macrosphenus flavicans and Sylvietta virens, which should be the root of the Macrosphenidae, occurred around 20.3 mya. If the Macrosphenidae weren't such a coherent group, I might think of dividing them. But there is no need for that.
- Yellow Longbill, Macrosphenus flavicans
- Kemp's Longbill, Macrosphenus kempi
- Gray Longbill, Macrosphenus concolor
- Pulitzer's Longbill, Macrosphenus pulitzeri
- Kretschmer's Longbill, Macrosphenus kretschmeri
- Rockrunner, Achaetops pycnopygius
- Cape Grassbird, Sphenoeacus afer
- Moustached Grass Warbler, Melocichla mentalis
- Victorin's Warbler, Cryptillas victorini
- Long-billed Crombec, Sylvietta rufescens
- Red-capped Crombec, Sylvietta ruficapilla
- Red-faced Crombec, Sylvietta whytii
- Somali Crombec, Sylvietta isabellina
- Green Crombec, Sylvietta virens
- Philippa's Crombec, Sylvietta philippae
- Lemon-bellied Crombec, Sylvietta denti
- Northern Crombec, Sylvietta brachyura
- White-browed Crombec, Sylvietta leucophrys
What about the Cisticolidae?
We've now reached the first real point of contention between the big three According to Oliveros et al. (2019) and Stiller et al. (2025), the Cisticola familiy (Cisticolidae) is the next branch, as shown on the diagram above. Kuhl et. (2021) found it to be the basal group in the next clade, containing the reed warblers (Acrocephalidae). This is an issue that's persisted. Fregin et al. (2012), which sampled more species from these groups (but fewer genes), also included Cisticolidae in the Acrocephalus clade, but as a basal group. Stiller et al. use more data per species, and Oliveros et al. include all of the relevant families. They agree that Cisticolidae is the next branch, and I'll go with them — for now.
Cisticolidae: Cisticolas Sundevall, 1872
26 genera, 158 species HBW-11
Oliveros et al. (2019) estimated the age of the Cisticolidae branch at about 23.1 mya, while Stiller et al. (2024) say 17.5 mya. Unfortunately, Kuhl et al. (2021) use a different topology, so their estimate doesn't apply to TiF. As always, there are error bars on these estimates, and a number like 20 mya is within both of them. Oliveros et al. also put ages on the Neomixinae and Priniinae / Cisticolinae split. They put the former split at around 19 mya and the later around 15.4 mya.
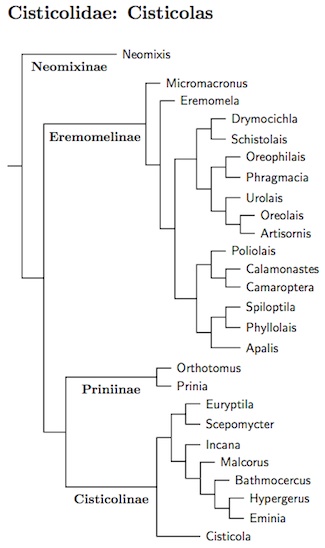 The cisticola family has grown since Sibley and Ahquist (1990) separated it
from the Sylviidae. Gains include
Eremomela,
Micromacronus (Oliveros et al., 2012),
Neomixis (except flavoviridis, now in Hartertula),
Phyllolais. Poliolais and most of Orthotomus
(except two species now in Phyllergates).
On the negative side, Rhopophilus has been lost to the
Paradoxornithidae (Alström et al., 2006) and two prinias have been put
in Laticilla and moved to the Pellorneidae (Olsson et al., 2013a).
Following Nguembock et al. (2008a), I've moved the
Black-collared Apalis and Ruwenzori Apalis to Oreolais, which is
sister to Artisornis. Like the Artisornis, the Oreolais
have 10 rather than 12 rectrices. Also, Mrs. Moreau's Warbler and
the recently split Rubeho Warbler are now listed in the genus
Scepomycter.
The cisticola family has grown since Sibley and Ahquist (1990) separated it
from the Sylviidae. Gains include
Eremomela,
Micromacronus (Oliveros et al., 2012),
Neomixis (except flavoviridis, now in Hartertula),
Phyllolais. Poliolais and most of Orthotomus
(except two species now in Phyllergates).
On the negative side, Rhopophilus has been lost to the
Paradoxornithidae (Alström et al., 2006) and two prinias have been put
in Laticilla and moved to the Pellorneidae (Olsson et al., 2013a).
Following Nguembock et al. (2008a), I've moved the
Black-collared Apalis and Ruwenzori Apalis to Oreolais, which is
sister to Artisornis. Like the Artisornis, the Oreolais
have 10 rather than 12 rectrices. Also, Mrs. Moreau's Warbler and
the recently split Rubeho Warbler are now listed in the genus
Scepomycter.
The arrangement of the genera is based primarily on Olsson et al. (2013a). Alström et al. (2006), Johansson et al. (2008b), and Nguembock et al. (2007, 2008a, 2012) were also consulted.
Olsson et al. found that monotypic genera Heliolais and Urorhipis are embedded within Prinia. They also found that two prinias do not belong within Prinia. They have been placed in Laticilla (Blyth 1845, type burnesii). The birds in question are the Rufous-vented Prinia, Laticilla burnesii, and the Swamp Prinia, Laticilla cinerascens. At present, we do not know which genus is the closest relative of Laticilla, but they are not part of Cisticolidae. They belong in Pellorneidae and seem to be closer to Pellorneum than to Graminicola.
The arrangement of the Orthotomus tailorbirds follows the estimated species tree in Sheldon et al. (2012). The Trilling Tailorbird, Orthotomus chloronotus, has been split from Philippine Tailorbird, Orthotomus castaneiceps as also advocated by Collar (2011). Further, I've added the newly discovered Cambodian Tailorbird, Orthotomus chaktomuk (Mahood et al, 2013). Amazingly, this new species was found right in Phnom Penh.
Neomixinae Olsson et al., 2013a
- Green Jery, Neomixis viridis
- Common Jery, Neomixis tenella
- Stripe-throated Jery, Neomixis striatigula
Eremomelinae Sharpe, 1883
- Visayan Miniature-Babbler, Micromacronus leytensis
- Mindanao Miniature-Babbler, Micromacronus sordidus
- Yellow-bellied Eremomela, Eremomela icteropygialis
- Salvadori's Eremomela, Eremomela salvadorii
- Yellow-vented Eremomela, Eremomela flavicrissalis
- Senegal Eremomela, Eremomela pusilla
- Green-backed Eremomela, Eremomela canescens
- Green-capped Eremomela, Eremomela scotops
- Karoo Eremomela, Eremomela gregalis
- Burnt-necked Eremomela, Eremomela usticollis
- Rufous-crowned Eremomela, Eremomela badiceps
- Turner's Eremomela, Eremomela turneri
- Black-necked Eremomela, Eremomela atricollis
- Red-winged Gray Warbler, Drymocichla incana
- Sierra Leone Prinia, Schistolais leontica
- White-chinned Prinia, Schistolais leucopogon
- Roberts's Warbler, Oreophilais robertsi
- Namaqua Warbler, Phragmacia substriata
- Green Longtail, Urolais epichlorus
- Black-collared Apalis, Oreolais pulcher
- Ruwenzori Apalis, Oreolais ruwenzorii
- Long-billed Forest-Warbler, Artisornis moreaui
- Red-capped Forest-Warbler, Artisornis metopias
- White-tailed Warbler, Poliolais lopezi
- Gray Wren-Warbler, Calamonastes simplex
- Miombo Wren-Warbler, Calamonastes undosus
- Stierling's Wren-Warbler, Calamonastes stierlingi
- Barred Wren-Warbler, Calamonastes fasciolatus
- Yellow-browed Camaroptera, Camaroptera superciliaris
- Olive-green Camaroptera, Camaroptera chloronota
- Green-backed Camaroptera, Camaroptera brachyura
- Hartert's Camaroptera, Camaroptera harterti
- Cricket Warbler, Spiloptila clamans
- Buff-bellied Warbler, Phyllolais pulchella
- Bar-throated Apalis, Apalis thoracica
- Yellow-throated Apalis, Apalis flavigularis
- Namuli Apalis, Apalis lynesi
- Taita Apalis, Apalis fuscigularis
- Rudd's Apalis, Apalis ruddi
- Yellow-breasted Apalis, Apalis flavida
- Lowland Masked-Apalis, Apalis binotata
- Mountain Masked-Apalis, Apalis personata
- Black-throated Apalis, Apalis jacksoni
- White-winged Apalis, Apalis chariessa
- Black-capped Apalis, Apalis nigriceps
- Black-headed Apalis, Apalis melanocephala
- Chirinda Apalis, Apalis chirindensis
- Chestnut-throated Apalis, Apalis porphyrolaema
- Kabobo Apalis, Apalis kaboboensis
- Chapin's Apalis, Apalis chapini
- Sharpe's Apalis, Apalis sharpii
- Buff-throated Apalis, Apalis rufogularis
- Kungwe Apalis, Apalis argentea
- Karamoja Apalis, Apalis karamojae
- Bamenda Apalis, Apalis bamendae
- Gosling's Apalis, Apalis goslingi
- Gray Apalis, Apalis cinerea
- Brown-headed Apalis, Apalis alticola
Priniinae Roberts, 1922
- Common Tailorbird, Orthotomus sutorius
- Rufous-tailed Tailorbird, Orthotomus sericeus
- Rufous-fronted Tailorbird, Orthotomus frontalis
- Dark-necked Tailorbird, Orthotomus atrogularis
- Cambodian Tailorbird, Orthotomus chaktomuk
- Ashy Tailorbird, Orthotomus ruficeps
- Olive-backed Tailorbird, Orthotomus sepium
- Philippine Tailorbird, Orthotomus castaneiceps
- Trilling Tailorbird, Orthotomus chloronotus
- Gray-backed Tailorbird, Orthotomus derbianus
- Yellow-breasted Tailorbird, Orthotomus samarensis
- White-eared Tailorbird, Orthotomus cinereiceps
- Black-headed Tailorbird, Orthotomus nigriceps
- Striated Prinia, Prinia crinigera
- Brown Prinia, Prinia polychroa
- Black-throated Prinia, Prinia atrogularis
- Hill Prinia, Prinia superciliaris
- Gray-crowned Prinia, Prinia cinereocapilla
- Rufous-fronted Prinia, Prinia buchanani
- Rufescent Prinia, Prinia rufescens
- Gray-breasted Prinia, Prinia hodgsonii
- Graceful Prinia, Prinia gracilis
- Jungle Prinia, Prinia sylvatica
- Bar-winged Prinia, Prinia familiaris
- Yellow-bellied Prinia, Prinia flaviventris
- Ashy Prinia, Prinia socialis
- Tawny-flanked Prinia, Prinia subflava
- Plain Prinia, Prinia inornata
- Pale Prinia, Prinia somalica
- River Prinia, Prinia fluviatilis
- Black-chested Prinia, Prinia flavicans
- Karoo Prinia, Prinia maculosa
- Drakensberg Prinia, Prinia hypoxantha
- Sao Tome Prinia, Prinia molleri
- Banded Prinia, Prinia bairdii
- Red-fronted Warbler, Prinia rufifrons
- Red-winged Warbler, Prinia erythroptera
Cisticolinae Sundevall, 1872
- Cinnamon-breasted Warbler, Euryptila subcinnamomea
- Winifred's Warbler, Scepomycter winifredae
- Rubeho Warbler, Scepomycter rubehoensis
- Socotra Warbler, Incana incana
- Rufous-eared Warbler, Malcorus pectoralis
- Black-headed Rufous Warbler, Bathmocercus cerviniventris
- Black-faced Rufous Warbler, Bathmocercus rufus
- Oriole Warbler, Hypergerus atriceps
- Gray-capped Warbler, Eminia lepida
- Red-faced Cisticola, Cisticola erythrops
- Singing Cisticola, Cisticola cantans
- Whistling Cisticola, Cisticola lateralis
- Trilling Cisticola, Cisticola woosnami
- Chattering Cisticola, Cisticola anonymus
- Bubbling Cisticola, Cisticola bulliens
- Chubb's Cisticola, Cisticola chubbi
- Hunter's Cisticola, Cisticola hunteri
- Black-lored Cisticola, Cisticola nigriloris
- Lazy Cisticola, Cisticola aberrans
- Rock-loving Cisticola, Cisticola emini
- Rattling Cisticola, Cisticola chiniana
- Boran Cisticola, Cisticola bodessa
- Churring Cisticola, Cisticola njombe
- Ashy Cisticola, Cisticola cinereolus
- Tana River Cisticola, Cisticola restrictus
- Tinkling Cisticola, Cisticola rufilatus
- Gray-backed Cisticola, Cisticola subruficapilla
- Wailing Cisticola, Cisticola lais
- Lynes's Cisticola, Cisticola distinctus
- Winding Cisticola, Cisticola marginatus
- Rufous-winged Cisticola, Cisticola galactotes
- Luapula Cisticola, Cisticola luapula
- Ethiopian Cisticola, Cisticola lugubris
- Coastal Cisticola, Cisticola haematocephalus
- Chirping Cisticola, Cisticola pipiens
- Carruthers's Cisticola, Cisticola carruthersi
- Levaillant's Cisticola, Cisticola tinniens
- Stout Cisticola, Cisticola robustus
- Aberdare Cisticola, Cisticola aberdare
- Croaking Cisticola, Cisticola natalensis
- Red-pate Cisticola, Cisticola ruficeps
- Dorst's Cisticola, Cisticola guinea
- Tiny Cisticola, Cisticola nana
- Short-winged Cisticola, Cisticola brachypterus
- Rufous Cisticola, Cisticola rufus
- Foxy Cisticola, Cisticola troglodytes
- Piping Cisticola / Neddicky, Cisticola fulvicapilla
- Long-tailed Cisticola, Cisticola angusticauda
- Black-tailed Cisticola, Cisticola melanurus
- Zitting Cisticola, Cisticola juncidis
- Socotra Cisticola, Cisticola haesitatus
- Madagascan Cisticola, Cisticola cherina
- Desert Cisticola, Cisticola aridulus
- Cloud Cisticola, Cisticola textrix
- Black-backed Cisticola, Cisticola eximius
- Dambo Cisticola, Cisticola dambo
- Pectoral-patch Cisticola, Cisticola brunnescens
- Pale-crowned Cisticola, Cisticola cinnamomeus
- Wing-snapping Cisticola, Cisticola ayresii
- Golden-headed Cisticola, Cisticola exilis
Acrocephalidae and allies
The next five families are relatively closely related. As mentioned above, the Cisticolidae may also belong here. Although it belongs in this clade, there's also some uncertainty about exactly where the Pnoepygidae go. Kuhl et al. (2021) place the cupwings (Pnoepygidae) sister to the the reed warblers (Acrocephalidae), as did Fregin et al. (2012). Neither Oliveros et al. (2019) nor Stiller et al. (2024) included the cupwings in their analyses.
Pnoepygidae: Cupwings Gelang et al., 2009
1 genus, 5 species Not HBW Family
Gelang et al. (2009) found that the Pnoepyga wren-babblers (now called cupwings) did not belong to any of the extant Sylvioidea families. They recommend placing it in its own family. Their analysis suggested they might be close to the cisticolas or bulbuls, but with low support. The overlapping analyses of Alström et al. (2011a) and Irestedt et al. (2011) included any family that might be related. The Pnoepygidae ended up as a basal member of this group. We follow Fregin et al. (2012) and Kuhl et al. (2021) in considering Pnoepygidae sister to Acrocephalidae.
The Chinese Cupwing, Pnoepyga mutica, has been split from the Scaly-breasted Cupwing, Pnoepyga albiventer based on Päckert et al. (2013a), which is why I list 5 cupwings rather than 4. Yes, it's a bit speculative.
- Nepal Cupwing, Pnoepyga immaculata
- Pygmy Cupwing, Pnoepyga pusilla
- Taiwan Cupwing, Pnoepyga formosana
- Scaly-breasted Cupwing, Pnoepyga albiventer
- Chinese Cupwing, Pnoepyga mutica
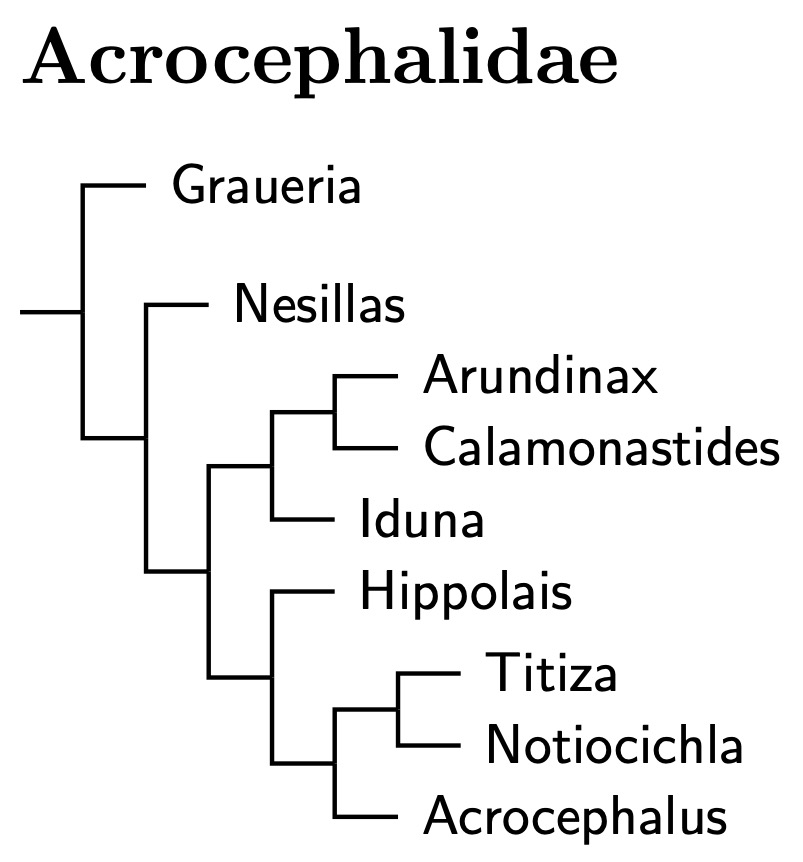
Acrocephalidae: Reed-Warblers Dresser, 1880 (1838)
9 genera, 64 species Not HBW Family
The reed-warblers, Acrocephalidae, are another new family formed out of the Sylviidae. The arrangement here is primarily based on Arbabi et al. (2014a) with help from Fleischer et al. (2007), Cibois et al. (2007, 2008, 2011) and Fregin et al. (2009). The multigene analysis of Arbabi supercedes much of Fregin et al. (2009), Leisler et al. (1997) and Helbig and Seibold (1999). Cibois et al. (2007, 2011) gave insight into how the reed-warblers expanded into Polynesia. The exact branching pattern differs some between Cibois et al. and Fleischer et al., and I have used a compromise version here which presumes each did best where taxon sampling was densest. I follow this in preference to Fregin et al. for the Polynesian species, as Fregin et al. only sampled one gene from that group. For the other species, Fregin et al. is given preference.
Graueria: Oliveros et al. (2019) seem to be the only ones to include Grauer's Warbler, Graueria vittata. They also included Malagasy Brush-Warbler, Nesillas typica, and Oriental Reed-Warbler, Acrocephalus orientalis. The smallest clade containing the latter two is what was previously considered Acrocephalidae. Graueria is sister to that.
Oliveros et al. dated the common ancestor of Graueria and the other Acrocephalidae at about 16.7 mya. That means it makes sense to think of them as belonging to the same family. Given the separation, we rank Graueria as a subfamily, Graueriinae. The Oliveros et al. estimates also indicate that the common ancestor of the remaining reed warblers lived less than 10 million years ago. Accordingly, I've put the remaining reed warblers in subfamily Acrocephalinae.
 |
| Click for Acrocephalidae tree |
|---|
Walking the Acrocephalid tree: Nesillas is basal in the subfamily Acrocephalinae. Both Johansson et al. (2008b) and Oliveros et al. (2019) included one member of Nesillas in their analysis. Both found that it was part of Acrocephalidae. Johansson et al. and Fregin et al. (2009) concur that it is also more basal among the acrocephalids sampled (they didn't include Graueria).
The single species in Calamonastides was formerly included in Chloropeta. Following Fregin et al. (2009), the other two former Chloropeta have been submerged in Iduna, which otherwise contains former Hippolais warblers. Earlier work suggested that the Thick-billed Warbler, Arundinax aedon, may not be closely related to the remaining Acrocephalus. Fregin et al. place it sister to Iduna, but Arbabi et al. (2014a) find it sister to Calamonastides.
Both Leisler et al. and Helbig and Seibold endorsed some of the traditional subgenera of Acrocephalus. Fregin et al. (2009) is consistent with this arrangement. The small streaked Acrocephalus become Titiza Billberg, 1828 (which has priority over Calamodus Kaup, 1829) while most of the small unstreaked Acrocephalus become Notiocichla. Although often placed elsewhere, the Large-billed Reed-Warbler belongs in Notiocichla (Bensch and Pearson, 2002). Iduna consists of former Hippolias warblers.
The Millerbird is sometimes considered as two species, Nihoa Millerbird, Acrocephalus kingi, and the extinct Laysan Millerbird, Acrocephalus familiaris. The genetic distance is in the species/subspecies borderline area (Fleischer et al., 2007). AOU currently treats them as one species, as I do here.
Cibois et al. (2011) were the first to include A. luscinius, syrinx, and rehsei in their analysis. They also examined a number of the extinct island taxa (see also Cibois et al., 2008). As a result, The Saipan Reed-Warbler, Acrocephalus hiwae, Pagan Reed-Warbler, Acrocephalus yamashinae, and Mangareva Reed-Warbler, Acrocephalus astrolabii are split from Nightingale Reed-Warbler, Acrocephalus luscinius. Also, Garrett's Reed-Warbler, Acrocephalus musae and Moorea Reed-Warbler, Acrocephalus longirostris are split from Tahiti Reed-Warbler, Acrocephalus caffer. Note that most of these taxa are now extinct.
Graueriinae: Grauer's Warbler Informal, Oliveros et al., 2019
- Grauer's Warbler, Graueria vittata
Acrocephalinae: Reed-Warblers Dresser, 1880 (1838)
- Malagasy Brush-Warbler, Nesillas typica
- Subdesert Brush-Warbler, Nesillas lantzii
- Anjouan Brush-Warbler, Nesillas longicaudata
- Grand Comoro Brush-Warbler, Nesillas brevicaudata
- Moheli Brush-Warbler, Nesillas mariae
- Aldabra Brush-Warbler, Nesillas aldabrana
- Thick-billed Warbler, Arundinax aedon
- Papyrus Yellow Warbler, Calamonastides gracilirostris
- Booted Warbler, Iduna caligata
- Sykes's Warbler, Iduna rama
- Olivaceous Warbler / Eastern Olivaceous-Warbler, Iduna pallida
- Isabelline Warbler / Western Olivaceous-Warbler, Iduna opaca
- African Yellow-Warbler, Iduna natalensis
- Mountain Yellow-Warbler, Iduna similis
- Upcher's Warbler, Hippolais languida
- Olive-tree Warbler, Hippolais olivetorum
- Melodious Warbler, Hippolais polyglotta
- Icterine Warbler, Hippolais icterina
- Sedge Warbler, Titiza schoenobaenus
- Aquatic Warbler, Titiza paludicola
- Moustached Warbler, Titiza melanopogon
- Speckled Reed-Warbler, Titiza sorghophila
- Black-browed Reed-Warbler, Titiza bistrigiceps
- Paddyfield Warbler, Notiocichla agricola
- Manchurian Reed-Warbler, Notiocichla tangorum
- Blunt-winged Warbler, Notiocichla concinens
- Blyth's Reed-Warbler, Notiocichla dumetorum
- Large-billed Reed-Warbler, Notiocichla orina
- Marsh Warbler, Notiocichla palustris
- African Reed-Warbler, Notiocichla baeticata
- Mangrove Reed-Warbler, Notiocichla avicenniae
- Eurasian Reed-Warbler, Notiocichla scirpacea
- Caspian Reed-Warbler, Notiocichla fusca
- Basra Reed-Warbler, Acrocephalus griseldis
- Lesser Swamp-Warbler, Acrocephalus gracilirostris
- Greater Swamp-Warbler, Acrocephalus rufescens
- Cape Verde Swamp-Warbler / Cape Verde Warbler, Acrocephalus brevipennis
- Seychelles Warbler, Acrocephalus sechellensis
- Madagascan Warbler / Madagascan Swamp-Warbler, Acrocephalus newtoni
- Rodrigues Warbler, Acrocephalus rodericanus
- Great Reed-Warbler, Acrocephalus arundinaceus
- Oriental Reed-Warbler, Acrocephalus orientalis
- Clamorous Reed-Warbler, Acrocephalus stentoreus
- Nightingale Reed-Warbler, Acrocephalus luscinius
- Saipan Reed-Warbler, Acrocephalus hiwae
- Australian Reed-Warbler, Acrocephalus australis
- Aguiguan Reed-Warbler, Acrocephalus nijoi
- Carolinian Reed-Warbler, Acrocephalus syrinx
- Kiritimati Reed-Warbler / Bokikokiko, Acrocephalus aequinoctialis
- Southern Marquesan Reed-Warbler, Acrocephalus mendanae
- Pagan Reed-Warbler, Acrocephalus yamashinae
- Nauru Reed-Warbler, Acrocephalus rehsei
- Millerbird, Acrocephalus familiaris
- Pitcairn Reed-Warbler, Acrocephalus vaughani
- Henderson Reed-Warbler, Acrocephalus taiti
- Cook Reed-Warbler, Acrocephalus kerearako
- Rimatara Reed-Warbler, Acrocephalus rimitarae
- Garrett's Reed-Warbler, Acrocephalus musae
- Tahiti Reed-Warbler, Acrocephalus caffer
- Moorea Reed-Warbler, Acrocephalus longirostris
- Northern Marquesan Reed-Warbler, Acrocephalus percernis
- Tuamotu Reed-Warbler, Acrocephalus atyphus
- Mangareva Reed-Warbler, Acrocephalus astrolabii
Donacobiidae: Donacobius Aleixo & Pacheco, 2006
1 genus, 1 species Not HBW Family
- Black-capped Donacobius, Donacobius atricapilla
The monotypic Black-capped Donacobius has variously been considered a wren, thrush, or mockingbird. It is none of these! It is a sylvioid that is sister to the Malagasy warblers (Bernieridae). The most recent common ancestor of the two groups is estimated by Oliveros et al. (2019) to have lived about 18.8 mya. The Donacobius is quite different from the Malagasy warblers. That and its age justify placing it in its own family.
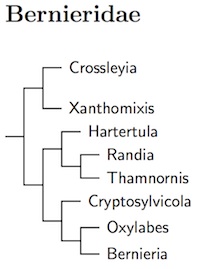
Bernieridae: Malagasy Warblers Cibois et al., 2010
8 genera, 12 species Not HBW Family
 |
| Click for Bernieridae species tree |
|---|
Most of the species in new Malagasy warbler family, Bernieridae, comes from the Sylviidae. The big exception is Bernieria itself, which comes from the Pycnonotidae (Cibois et al., 1999, 2001).
Block (2012) confirmed that Rand's Warbler belongs in the Bernieridae. The arrangement here is based on Block (2012), who shows that the Dusky Tetraka, formerly Xanthomixis tenebrosa, belongs in Crossleyia as Crossleyia tenebrosa. Block (2012) split Bernieria inceleber, which I'm calling Pale Bernieria, from Long-billed Bernieria, Bernieria madagascariensis. He also found evidence of another cryptic species in this complex, which is yet to be described.
- Madagascan Yellowbrow, Crossleyia xanthophrys
- Dusky Tetraka, Crossleyia tenebrosa
- Spectacled Tetraka, Xanthomixis zosterops
- Gray-crowned Tetraka, Xanthomixis cinereiceps
- Appert's Tetraka, Xanthomixis apperti
- Wedge-tailed Jery, Hartertula flavoviridis
- Rand's Warbler, Randia pseudozosterops
- Kiritika Warbler / Thamnornis, Thamnornis chloropetoides
- Cryptic Warbler, Cryptosylvicola randrianasoloi
- White-throated Oxylabes, Oxylabes madagascariensis
- Pale Bernieria, Bernieria inceleber
- Long-billed Bernieria, Bernieria madagascariensis
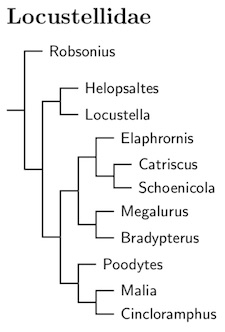
Locustellidae: Grassbirds Bonaparte, 1854
11 genera, 63 species Not HBW Family
The grassbirds are another new family, Locustellidae. As with several of the other new families, the Locustellidae are comprised entirely of former Sylviidae. Indeed, many were members of the Megalurinae subfamily. As a result, the name Megaluridae has been used for this family. This is a mistake. It fails to account for the addition of Locustella to the group. The name Locustellidae has priority over Megaluridae. Locustellidae was named by Bonaparte in 1854, while Megaluridae dates to Blyth (1875, p.117; see also Bock, 1994).
 |
| Click for Locustellidae species tree |
|---|
For a long time, both Megalurus and Locustella have been included in families with more senior names (e.g. Sylviidae, Turdidae, Muscicapidae). Megaluridae appears to have next seen the light of day (as Megalurinae) in Sibley and Ahlquist (1985a). As their Megalurinae did not contain Locustella, their use has no bearing on the correct name of this family as currently constituted. Alström et al. (2006) added Locustella to the Megaluridae, but it apparently did not occur to them that Locustella might bring its own family name with it. This was subsequently followed by a few authors (and this website), but Sangster et al. (2009) listed the family as Locustellidae. A little checking showed it had priority, and this website now follows their lead.
Former Babblers: Oliveros et al. (2012) found that two additional genera previously regarded as babblers also belong with the grassbirds: Robsonius and Malia. A little denser taxon sampling would have been nice, but Robsonius appears to be basal, and Malia appears to be somewhere near Cincloramphus. Alström et al. (2018) clarified the position of these taxa, with Robsonius basal and Malia sister to an expanded Cincloramphus.
Grassbird Phylogeny
The Asian Bradypterus species were previously moved to Locustella using a combination of Drovetski et al (2004), guesswork based on geography and rumors of a paper by Alström and others (which I had not then seen). Alström et al. (2011b) confirmed the previous restructuring of Locustella and Bradypterus. The more recent paper by Alström et al. (2018a) found that the Bamboo Warbler also belongs in Locustella rather than Bradypterus. This is something of a surprise as it is an African species. Six species from NE Asia are rather distantly related to the rest of Locustella, and have been separated as the new genus Helopsaltes Alström et al. 2018a (type species certhiola).
The Marsh Grassbird, now Helopsaltes pryeri, was originally moved from Megalurus to Locustella based on Drovetski et al. (2004b). This was confirmed by Alström et al. (2011b, 2018a). It is part of the NE Asian clade moved to Helopsaltes.
The second big group has also been restructed based on Alström et al. (2018a). The Fan-tailed Grassbird moves from Schoenicola to Catriscus, while the Bristled Grassbird (Chaetornis) has been merged into Schoenicola.
Alström et al. (2011b) found that the Striated Grassbird, Megalurus palustris, didn't seem to group with the rest of Megalurus. Since it is the type species, that means the others need to change their genus, and have now been moved into Poodytes and Cincloramphus (some were previously placed in Bowdleria).
The genus Dromaeocercus was previously merged into Bradypterus. Following Alström et al. (2018a), Amphilais has also been merged into Bradypterus.
The other big group is comprised of three genera: Poodytes, Malia, and Cincloramphus. Poodytes has been expanded to include Eremiornis, and Buettikoferella and Megalurulus have been merged into Cincloramphus. The Tawny Grassbird has also joined Cincloramphus.
Splits, Lumps, and New Species
Hosner et al. (2013c) found a previously undiscovered Robsonius species, the Sierra Madre Ground-Warbler Robsonius thompsoni.
Drovetski et al. (2004) and Alström et al. (2011b) found substantial genetic differences between the Sakhalin Grasshopper-Warbler, Locustella amnicola, and Gray's Grasshopper-Warbler, Locustella fasciolata. These had been suspected to be separate species, and so are split here.
Alström et al. (2015b) described a new species, the Sichuan Bush-Warbler, Locustella chengi. They also undertook a reevaluation of the Russet Bush-Warbler complex, including subspecies alishanensis, chengi, idonea, mandelli, melanorhyncha, montis, seebohmi, and timorensis. Based on morphology, song, genetics, and biogoegraphy, they recommended lumping Timor Bush-Warbler, Locustella timorensis, into Javan Bush-Warbler, Locustella montis and elevating idonea to species status. They did not provide an English name, but IOC has picked Dalat Bush-Warbler for Locustella idonea. I would have preferred a name that better reflects its range, such as Tay Nguyen Bush-Warbler (after the Central Highlands, Tây Nguyên). They obtained two different phylogenies for the complex. I've adopted the BEAST chronogram, but it may not be entirely correct.
Alström et al. (2011b) also found that the Little Rush Warbler, Bradypterus baboecala, consists of at least two species. The Highland Rush Warbler, Bradypterus centralis, has been split from it. The correct allocation of subspecies remains uncertain. Alström et al. examined 4 subspecies: transvaalensis and tongensis from the baboecala group and centralis and elgonensis from the centralis group. IOC has included only the two subspecies in centralis, while H&M-4 (Dickinson and Christidis, 2014) also included chadensis and sudanensis in the Highland Rush Warbler (B. centralis).
There is additional information available. Kennerley and Pearson (2010) note that some birds from Cameroon, usually thought to be centralis have vocalizations that “sound like southern birds rather than those of SW Uganda and Rwanda.” They suggest that these birds are not centralis. Since then, they have been considered part of msiri. Stervander et al. (2005) found Rush Warbler on the Jos Plateau in central Nigeria that responded to playback of Little Rush Warbler calls, even though it looked more like centralis. Dowsett and Dowsett-Lemaire (2015) found that birds at Lake Awassa in Ethiopia responded to playback of songs from South Africa, suggesting that abyssinicus belongs in the baboecala group. More work needs to be done to properly sort out this situation.
- Sierra Madre Ground-Warbler, Robsonius thompsoni
- Bicol Ground-Warbler / Gray-banded Babbler, Robsonius sorsogonensis
- Cordillera Ground-Warbler / Rusty-faced Babbler, Robsonius rabori
- Gray's Grasshopper-Warbler, Helopsaltes fasciolata
- Sakhalin Grasshopper-Warbler, Helopsaltes amnicola
- Marsh Grassbird, Helopsaltes pryeri
- Pallas's Grasshopper-Warbler, Helopsaltes certhiola
- Styan's Grasshopper-Warbler, Helopsaltes pleskei
- Middendorff's Grasshopper-Warbler, Helopsaltes ochotensis
- Lanceolated Warbler, Locustella lanceolata
- Bamboo Warbler, Locustella alfredi
- River Warbler, Locustella fluviatilis
- Savi's Warbler, Locustella luscinioides
- Brown Bush-Warbler, Locustella luteoventris
- Chinese Bush-Warbler, Locustella tacsanowskia
- Common Grasshopper-Warbler, Locustella naevia
- Long-billed Bush-Warbler, Locustella major
- Friendly Bush-Warbler, Locustella accentor
- Long-tailed Bush-Warbler, Locustella caudata
- Chestnut-backed Bush-Warbler, Locustella castanea
- Baikal Bush-Warbler, Locustella davidi
- Spotted Bush-Warbler, Locustella thoracica
- West Himalayan Bush-Warbler, Locustella kashmirensis
- Taiwan Bush-Warbler, Locustella alishanensis
- Benguet Bush-Warbler, Locustella seebohmi
- Javan Bush-Warbler, Locustella montis
- Sichuan Bush-Warbler, Locustella chengi
- Russet Bush-Warbler, Locustella mandelli
- Dalat Bush-Warbler, Locustella idonea
- Sri Lanka Bush-Warbler, Elaphrornis palliseri
- Fan-tailed Grassbird, Catriscus brevirostris
- Bristled Grassbird, Schoenicola striatus
- Broad-tailed Grassbird, Schoenicola platyurus
- Striated Grassbird, Megalurus palustris
- Knysna Warbler, Bradypterus sylvaticus
- Bangwa Forest-Warbler, Bradypterus bangwaensis
- Cinnamon Bracken Warbler, Bradypterus cinnamomeus
- Evergreen Forest-Warbler, Bradypterus lopezi
- Barratt's Warbler, Bradypterus barratti
- Gray Emutail, Bradypterus seebohmi
- Brown Emutail, Bradypterus brunneus
- Little Rush Warbler, Bradypterus baboecala
- White-winged Swamp Warbler, Bradypterus carpalis
- Highland Rush Warbler, Bradypterus centralis
- Ja River Scrub Warbler, Bradypterus grandis
- Grauer's Swamp Warbler, Bradypterus graueri
- Spinifex-bird, Poodytes carteri
- Fly River Grassbird, Poodytes albolimbatus
- Little Grassbird, Poodytes gramineus
- Chatham Fernbird, Poodytes rufescens
- New Zealand Fernbird, Poodytes punctatus
- Malia, Malia grata
- Brown Songlark, Cincloramphus cruralis
- Rusty Thicketbird, Cincloramphus rubiginosus
- New Britain Thicketbird, Cincloramphus grosvenori
- Buff-banded Thicketbird, Cincloramphus bivittatus
- Rufous Songlark, Cincloramphus mathewsi
- Tawny Grassbird, Cincloramphus timoriensis
- Papuan Grassbird, Cincloramphus macrurus
- Bougainville Thicketbird, Cincloramphus llaneae
- Melanesian Thicketbird, Cincloramphus whitneyi
- New Caledonian Thicketbird, Cincloramphus mariae
- Long-legged Thicketbird, Cincloramphus rufus